 
The smaller the difference, the “happier” (more stable) the atom is. The purpose of formal charges is to compare the difference between the number of valence electrons in the free atom and the number of electrons the atom “owns” when it is bonded. 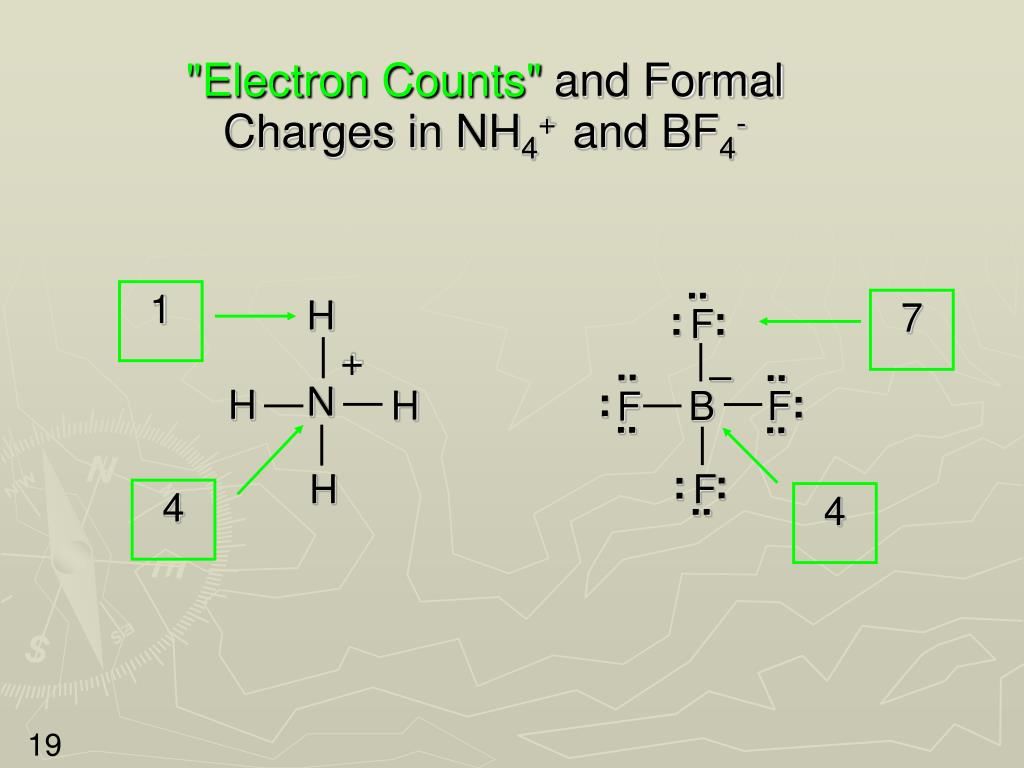
Since the two oxygen atoms have the same bonding, one calculation is enough for both oxygen atoms.ġ.2.3 Guidelines about Formal Charges in Lewis Structures Formal charges check: all atoms have formal charges equals to 0 in this structure. This is the complete Lewis structure of CO 2.įor Lewis structure purposes, the lone-pairs can only be moved f rom terminal atoms to the central atom to form multiple bonds, not the other way around.ħ. Moving one lone pair from each terminal O atom, the following structure is obtained. The central C atom does not get octet yet, we should do next step.Ħ. It is very important to keep in mind that the remaining electrons should be used to give the octet of terminal atoms first!ĥ. The remaining 12 electrons must be used to complete the octet for both terminal O atoms first, and no electrons left after that. Four electrons are used so far, and there are 16 – 4 = 12 electrons remained.Ĥ. Total number of valence electrons: 4 (C atom) + 2×6 (2 O atoms) = 16Īlways DOUBLE CHECK: In the correct Lewis structure, the total number of electrons involved (bonding plus non-bonding electrons) must be equal to this number, less or more are both incorrect!!Ĭarbon atoms are always central, so the skeletal structure is: O - C - Oģ. Calculate the Formal Charges on all atoms and label the non-zero formal charges in the structure:įormal Charge on an atom = No. If not, then complete the octets of all central atoms by moving lone pairs from terminal atoms to form multiple bonds.ħ. If you have used up all the valence electrons to complete octets for all the atoms, you are done.Ħ. Using the remaining valence electrons, complete the octets of the terminal atoms first, then complete as many as possible for the central atoms.ĥ. 
For each single bond, subtract two electrons from the total number of valence electrons.Ĥ. Central atoms are generally those with the lowest EN.ī) Connect the central atom with each of the terminal atoms by drawing a single bond.ģ.Write a plausible skeletal structure using the following steps:Ī) Write atomic symbols for the central and terminal atoms. The total number of electrons = 5 (N atom) + 4×1 (four H atoms) -1 (minus the charge for cation) = 8 valence electronsĢ. For ions, make sure charges are properly included in the calculation. Calculate the total number of valence electrons. Lewis Structure Drawing Procedu re for Polyatomic Molecules and Ionsġ. It is very important that you use the following procedure to get the correct Lewis structures for polyatomic molecules and ions. A specific procedure with certain steps has to be followed. Draw the Lewis structure of the N 2 molecule.ġ.2.2 Lewis Structures of Polyatomic Molecules or Ionsįor more complicated polyatomic molecules and ions, the Lewis structures cannot be obtained by simply combining Lewis symbols. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
